Anti-Human Epidermal Growth Factor Receptor (EGFR)
Anti-Human Epidermal Growth Factor Receptor (EGFR)
Product No.: E100
- -
- -
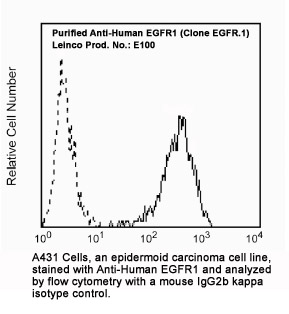
Clone EGFR.1 Target Epidermal Growth Factor Receptor Formats AvailableView All Product Type Monoclonal Antibody Alternate Names ERBB, ERBB1, mENA Isotype Mouse IgG2b κ Applications FC |
Data
- -
- -
Antibody DetailsProduct DetailsReactive Species Human Host Species Mouse Immunogen A431 cultured cells Product Concentration 0.5 mg/ml Formulation This purified antibody is formulated in 0.01 M phosphate buffered saline (150 mM NaCl) PBS pH 7.4, 1% BSA and 0.09% sodium azide as a preservative. Storage and Handling This purified antibody is stable when stored at 2-8°C. Do not freeze. Country of Origin USA Shipping Next Day Ambient RRIDAB_2829966 Each investigator should determine their own optimal working dilution for specific applications. See directions on lot specific datasheets, as information may periodically change. DescriptionDescriptionSpecificity Anti-Human EGFR recognizes the (Mr 170 kDa) extracellular domain of the epidermal growth factor receptor on many cell surfaces. The EGFR mediates the initial response of cells to EGF. It has an extracellular region which binds EGF and an intracellular region which exhibits tyrosine kinase activity.1 The receptor phosphorylates a number of protein substrates in addition to mediating autophosphorylation of tyrosine residues located near its carboxyl terminus.2,3 Background The epidermal growth factor receptor (EGFR; ErbB-1; HER1 in humans) is the cell-surface receptor for members of the epidermal growth factor family (EGF-family) of extracellular protein ligands. The epidermal growth factor receptor is a member of the ErbB family of receptors, a subfamily of four closely related receptor tyrosine kinases: EGFR (ErbB-1), HER2/c-neu (ErbB-2), Her 3 (ErbB-3) and Her 4 (ErbB-4). Mutations affecting EGFR expression or activity could result in cancer. EGFR (epidermal growth factor receptor) exists on the cell surface and is activated by binding of its specific ligands, including epidermal growth factor and transforming growth factor α (TGFα) (note, a full list of the ligands able to activate EGFR and other members of the ErbB family is given in the ErbB article). ErbB2 has no known direct activating ligand, and may be in an activated state constitutively or become active upon heterodimerization with other family members such as EGFR. Upon activation by its growth factor ligands, EGFR undergoes a transition from an inactive monomeric form to an active homodimer - although there is some evidence that preformed inactive dimers may also exist before ligand binding. In addition to forming homodimers after ligand binding, EGFR may pair with another member of the ErbB receptor family, such as ErbB2/Her2/neu, to create an activated heterodimer. There is also evidence to suggest that clusters of activated EGFRs form, although it remains unclear whether this clustering is important for activation itself or occurs subsequent to activation of individual dimers. Research Area Cell Biology . Immunology . Innate Immunity . Neuroscience . Synaptic Biology References & Citations1. Berger, SM. et al. (1987) J. of Pathology 152:297 2. Downward, J. et al. (1984) Nature 311:483 3. Gullick, WJ. et al. (1985) EMBO J. 4:2869 4. Gullick, WJ. et al. (1986) Cancer Research 46:285 5. Gullick, WJ. et al. (1991) Br. Med. Bulletin 47:87 Technical ProtocolsCertificate of Analysis |
Formats Available
- -
- -
Prod No. | Description |
|---|---|
E333 | |
E325 | |
E326 | |
E327 | |
E329 | |
E331 | |
E336 | |
E101 | |
E337 | |
E100 |